
・Letter to the Editor ・ Current
Issue IF in JCR CiteScore ・Submission・ In Press Recent Accepted PMC RSS
・・
Citation: Sheng Y, Sun W, Gu YS. Spectral-domain optical coherence tomography
dynamic changes and steroid response in multiple evanescent white dot syndrome.
Int J Ophthalmol 2017;10(8):1331-1333
Spectral-domain optical coherence tomography
dynamic changes and steroid response in multiple evanescent white dot syndrome
Yan Sheng, Wen Sun, Yang-Shun Gu
Department of Ophthalmology, the First Affiliated Hospital, College of
Medicine, Zhejiang University, Hangzhou 310003, Zhejiang Province, China
Correspondence to: Yang-Shun Gu.
Department of Ophthalmology, the First Affiliated Hospital, College of
Medicine, Zhejiang University, No.79 Qingchun Road, Hangzhou 310003, Zhejiang
Province, China. guyangshun @163.com
Received: 2017-03-01 Accepted: 2017-05-20
DOI:10.18240/ijo.2017.08.24
Citation: Sheng Y, Sun W, Gu YS. Spectral-domain optical coherence tomography
dynamic changes and steroid response in multiple evanescent white dot syndrome.
Int J Ophthalmol 2017;10(8):1331-1333
Dear
Editor,
Multiple
evanescent white dot syndrome (MEWDS) was first described in 1984 as a rare,
acute, unilateral, multifocal retinochoroidal disorder, typically affecting
young myopic women[1]. Previous studies with
fluorescein angiography (FA) and electrophysiology suggested that MEWDS to be a
disease in the retinal pigment epithelium (RPE) or outer retina[2], while recent studies with spectral-domain optical
coherence tomography (SD-OCT) suggested it may be an outer retinal disease due
to observation of hyperreflective material in outer retina and subtle
disruptions of the ellipsoid zone without RPE disruption[3].
However, some studies with indocyanine green angiography (ICGA) and choroidal
thickness measurement suggested that it may be a choroidal vascular or
inflammation disease[4]. It has not been clearly
demonstrated in the literatures that 1) what is the sequence of recovery for
the hyperreflectant materials and the disrupted ellipsoid zone; 2) whether
sub-lesion choroidal thickness changes sensitively during the process of the
disease; 3) what is the role of short term steroid in accelerating the
recovery. Here we report a MEWDS case with optical coherence tomography (OCT)
findings of an initial resolution of the hyperreflectant materials preceding
ellipsoid zone, with dynamic changes of sub-lesion choroidal thickness, and a
fast recovery after short-term median dose steroid therapy.
A
27-year-old health man presented with blurry vision and photopsia in his left
eye for two days. He has some flu-like symptoms 2wk earlier. His ocular history
was significant for amplyopia in his right eye. He wore contact lenses
measuring -11 DS OD and -9.5 DS OS. We observed the recovery process in this
case with ophthalmoscopy, SD-OCT, FA, ICGA, and Humphrey perimetry
examinations.
At
the first visiting, best-corrected visual acuities (BCVA) were 20/200 OD and
20/60 OS. Slit-lamp examination was unremarkable. Fundus examination showed
multiple gray-white, punctate dots located from posterior pole to the
midperipheral region, and several Fuchs
spots in nasal fundus and peripapillary atrophy
OS (Figure 1B). There was an irregular optic nerve with peripapillary
atrophy, some lacquercracks and Fuchs
spots at the posterior pole OD (Figure 1A). SD-OCT showed some
disruptions of the ellipsoid zone with hyperreflective material resting on the
RPE and extending toward the inner retina through interdigitation zone, ellipsoid
zone and outer nuclear layer which correlated to the white dot lesions observed
by ophthalmoscopy. Humphrey 30-2 visual field test revealed a marked enlarged
blind spot with central scotoma (Figure 2A). Because FA cannot be performed at
his first visit, the patient was scheduled this test on his next visit.
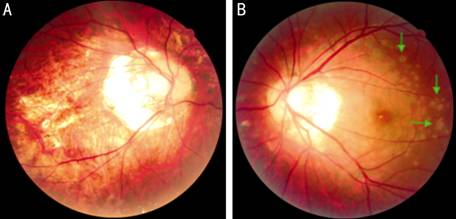
Figure
1 Color photography in baseline A: Irregular optic nerve with peripapillary atrophy,
some lacquercracks and Fuchs spots at the
posterior pole OD; B: Multiple gray-white, punctate dots located
lesions (green arrows), several Fuchs
spots in nasal fundus and peripapillary atrophy
OS.
Based
on the clinical findings and ancillary testing results, the patient was diagnosed
MEWDS. Due to the low vision in his right eye, the patient wanted a quick
recovery for the left eye to resume normal work and life. The patient,
therefore, was prescribed methylprednisolone 40 mg once daily for 7d.
At
his 1-week follow-up visit, the patient reported that the left vision improved
remarkably and photopsias had resolved completely. The BCVA increased to 20/25
OS. Dilated fundus examination showed resolution of most white dot lesions. The
visual field recovered remarkably (Figure 2B). FA demonstrated punctate
hyperfluorescent in early stage and minimally staining in late stage (Figure
3A, 3B). ICGA showed some hypofluorescent spots in late phase, which were
corresponded to the white dot lesions (Figure 3D). SD-OCT showed all the hyperreflective
material disappeared but disruption of ellipsoid zone in the corresponding
parts still exist (Figure 2B).

Figure
2 Changes in fundus, OCT and visual field
A: Baseline: color photography showed multiple gray-white,
punctate dots located lesions; OCT showed disruption of the ellipsoid zone and
some hyperreflective lesions (green arrows); visual field test revealed a
marked enlarged blind spot with central scotoma; B: One-week follow-up: color
photography showed white dot lesions gradually resolved; OCT showed
hyperreflective lesions disappeared but disruption of ellipsoid zone still
exist; visual field defects gradually resolved; C: One-month follow-up: color
photography, OCT and visual field are basically normal.
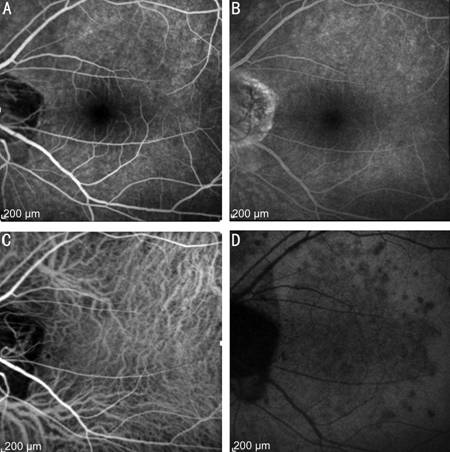
Figure
3 FA and ICGA A: Punctate
hyperfluorescent in early phase of FA; B: Minimally staining in late phase of
FA; C: No distinct abnormal finding in early phase of ICGA; D: Some
hypofluorescent lesions at the posterior pole in late phase of ICGA.
At
his 1-month follow-up visit, the patient reported that his left vision acuity
resolved completely, with BCVA of 20/20 OS. SD-OCT showed interdigitation zone,
ellipsoid zone and outer nuclear layer completely recovered. Repeated visual
field showed a mildly enlarged blind spot (Figure 2C).
Ten
lesions on OCT were selected for choroidal thickness measurement on each visit.
We performed a comparison of the choroidal thickness of the same ten lesions
among three visit times (follow up scan mode) (Figure 2). Statistical analyses
were performed with PASW 18.0 software. The mean choroidal thicknesses were
261.5 (±97.5) microns at the baseline, 228.2 (±90.1) microns at the 1-week
visit and 210.2 (±85.9) microns at the 4-week visit. The choroidal thickness of
the ten lesions was thicker at onset than that at the 1-week visit (P=0.012)
or that at the 4-week visit (P=0.001). However, the difference between 1
and 4wk visit was not statistically significant (P=0.052).
With
the widespread clinical use of OCT, retinal changes of MEWDS in OCT have been
reported in recent years. Diffuse disruptions of ellipsoid zone without RPE
disruption were reported in previous studies[3,5]. In addition to ellipsoid zone disruptions, Marsiglia et
al[3] found some protrusions of the hyperreflectant
material from the ellipsoid layer toward the outer nuclear layer correspond to
the location of dots seen with photography, ICGA, and FA in MEWDS. All the
retinal changes in OCT in the present case at the onset were consistent with
this literature reports. At the same time, we found resolution of hyperreflective
material occurred earlier than recovery of ellipsoid zone, which has not been
reported in previous literature. The resolution of hyperreflective materials
were corresponded to the regression of white dot lesions and with the clinical
improvement in visual acuity and visual field.
Regard
to choroidal thickness change of MEWDS in OCT, it just has been reported in a
few studies[3-5]. In those
studies, choroidal thickness was thicker in the acute phase and decreased
slightly in the convalescent phase. However, one report found the difference
between acute phase and convalescent phase was not statistically significant[3]. Furthermore, in all these studies, only subfoveal
choroidal thickness has been measured and compared[3-5]. In present case, ten lesions on OCT were selected for
choroidal thickness measurement and the choroidal thickness of beneath ten
lesions was thicker at onset than that in the convalescent phase (P=0.001).
To the best of our knowledge, there are no reports discussing the choroidal
thickness change in MEWDS with multiple lesions. We speculated that choroidal
thickening beneath the lesion in acute phase may represent dilated
choroicapillaries resulting from choroidal inflammation or ischemia, which
corresponded to finding in ICGA. Measuring the choroidal thickness just beneath
the lesions may be a more sensitive method to study choroicapillary changes in
MEWDS.
Our
patient was interesting in its faster recovery process. As MEWDS is a
self-resolving condition, Marsiglia et al[3]
observed 34 MEWDS patients without treatment and found the mean interval was
10wk for visual recovery and
Lombardo[2] reported visual acuity restoration and
clinical findings resolution in MEWDS without treatment are usually noted after
6 to 10wk. In an en-face OCT research in MEWDS, at 6-month follow-up,
incomplete recovery of the ellipsoid zone were observed in all the 4 patients
who have not been treated. Thus, the research suggested a possibility that a
corticosteroid treatment could have allowed larger recovering in the
photoreceptors integrity[6]. A case reported by
Takahashi et al[7] seems to support this
possibility; in that case, the visual acuity increased from 20/400 to 20/25
within 3d of the steroid pulse therapy (3000 mg for 3d). In the present
patient, immediately after the median dose steroid therapy, the visual acuity
of left eye increased from 20/60 to 20/25, the visual field recovered
remarkably, white dot lesions disappeared completely and choroidal thickness
decreased. This treatment result might suggest that steroid therapy may have a
hope of promoting an early recovery from MEWDS. Although the etiology of MEWDS
remains elusive, immunologic and infectious theories have been proposed[2]. According to a recent report, the
presenting signs of MEWDS may link to activation of the microglia or dilation
of the deep retinal capillary by the inflammation[3].
Due to the inflammatory process in MEWDS, steroid therapy may be an
effective treatment to promote the regression of the lesion and shorten the
course of MEWDS. Compared with pulse steroid therapy, medium-dose steroid was a
relatively safe treatment with a low risk of side effect and complication.
Thus, short term medium dose steroid treatment may be a choice to be applied in
limited situation in which patients with rapid decline in vision acuity and
need for a quick vision recovery to resume normal daily life and work like that
of the present patient.
ACKNOWLEDGEMENTS
Conflicts
of Interest: Sheng Y, None; Sun W, None; Gu YS,
None.
REFERENCES
1 Jampol LM, Sieving PA,
Pugh D, Fishman GA, Gilbert H. Multiple evanescent white dot syndrome. I.
Clinical findings. Arch Ophthalmol 1984;102(5):671-674.
[CrossRef] [PubMed]
2 Lombardo J. Multiple
evanescent white dot syndrome and acute zonal occult outer retinopathies. Optom Vis Sci 2003; 80(10):673-680. [CrossRef]
3 Marsiglia M,
Gallego-Pinazo R, Cunha de Souza E, Munk MR, Yu S, Mrejen S, Cunningham ET Jr,
Lujan BJ, Goldberg NR, Albini TA, Gaudric A, Francais C, Rosen RB, Freund KB,
Jampol LM, Yannuzzi LA. Expanded clinical spectrum of multiple evanescent white
dot syndrome with multimodal imaging. Retina
2016;36(1):64-74. [CrossRef] [PubMed]
4 Aoyagi R, Hayashi T, Masai
A, Mitooka K, Gekka T, Kozaki K, Tsuneoka H. Subfoveal choroidal thickness in
multiple evanescent white dot syndrome. Clin
Exp Optom 2012;95(2):212-217. [CrossRef] [PubMed]
5 Hua R, Chen K, Liu LM, Liu
NN, Chen L, Teng WP. Multi -modality imaging on multiple evanescent white dot
syndrome-a spectralis study. Int J
Ophthalmol 2012;5(5):644-647. [PMC free
article] [PubMed]
7 Takahashi Y, Ataka S, Wada
S, Kohno T, Nomura Y, Shiraki K. A case of multiple evanescent white dot
syndrome treated by steroid pulse therapy. Osaka
City Med J 2006;52(6):83-86. [PubMed]