IF in JCR CiteScore
Rank About IJO Current
Issue Featured Articles Articles In Press Recent Accepted
 International Journal
of Ophthalmology
International Journal
of Ophthalmology
2017; 10(9): 1436-1445
・Meta-Analysis・
Clinical
outcomes of small incision lenticule extraction versus femtosecond
laser-assisted LASIK for myopia: a Meta-analysis
Huan Yan1, Li-Yan Gong1, Wei
Huang1, Yan-Li Peng2,3
1Department of Ophthalmology, the
Second Affiliated Hospital of Chongqing Medical University, Chongqing Medical
University, Chongqing 400010, China
2Department of Ophthalmology,
Chongqing Aier-Mega Eye Hospital, Aier Eye Hospital Group, Chongqing 400060,
China
3Aier School of Ophthalmology,
Central South University, Changsha 410000, Hunan Province, China
Correspondence to: Yan-Li Peng.
Department of Ophthalmology, Chongqing Aier-Mega Eye Hospital, Aier Eye
Hospital Group, Chongqing 400060, China. ysl818106@sina.cn
Received: 2017-04-25
Accepted: 2017-06-07
Abstract
AIM: To evaluate the possible
differences in visual quality between small incision lenticule extraction
(SMILE) and femtosecond laser in situ keratomileusis (FS-LASIK) for myopia.
METHODS: A Meta-analysis was
performed. Patients were from previously reported comparative studies treated
with SMILE versus FS-LASIK. The PubMed, EMBASE, Cochrane, Web of Science and
Chinese databases (i.e. WANFANG and CNKI) were searched in Nov. of 2016 using
RevMan 5.1 version software. The differences in visual acuity, aberration and
biomechanical effects within six months postoperatively were showed.
Twenty-seven studies including 4223 eyes were included.
RESULTS: No significant
differences were observed between SMILE and FS-LASIK in terms of the proportion
of eyes that lost one or more lines of corrected distance visual acuity after
surgery (P=0.14), the proportion of eyes achieving an uncorrected distance
visual acuity of 20/20 or better (P=0.43), the final refractive spherical
equivalent (P=0.89), the refractive spherical equivalent within ±1.00 diopter
of the target values (P=0.80), vertical coma (P=0.45) and horizontal coma (P=0.06).
Compared with the FS-LASIK group, total higher-order aberration (P<0.001)
and spherical aberration (P<0.001) were higher and the decrease in corneal
hysteresis (P=0.0005) and corneal resistance factor (P=0.02) were lower in the
SMILE group.
CONCLUSION: SMILE and FS-LASIK
are comparable in efficacy, safety and predictability for correcting myopia.
However, the aberration in the SMILE group is superior to that in the FS-LASIK
group, and the loss of biomechanical effects may occur less frequently after
SMILE than after FS-LASIK.
KEYWORDS: Meta-analysis; small
incision lenticule extraction; femtosecond laser in situ keratomileusis; myopia
Citation: Yan H,
Gong LY, Huang W, Peng YL. Clinical outcomes of small incision lenticule
extraction versus femtosecond laser-assisted LASIK for myopia: a Meta-analysis.
Int J Ophthalmol 2017;
10(9):1436-1445
INTRODUCTION
Femtosecond laser in situ
keratomileusis (FS-LASIK) has been the most common corneal refractive surgery
and has proved to be effective, safe and predictable for treating myopia[1]. However, there is some problems to limit the application of
FS-LASIK, which includes the risk of flap-related complications and dry eye[2-3].
Small incision lenticule
extraction (SMILE) becomes a new option for myopic patients and the corneal
flap production is replaced by removing the corneal stroma lenticule from a
minimized incision to reduce the complications of corneal flap and dry eye
since 2011[4-5]. Recent studies have
indicated that there is less impairment of the biomechanical effects and more
corneal nerves are preserved when treated with SMILE compared with FS-LASIK
because of the complement of the anterior cornea, which can reduce the
incidence of dry eye[6-8]. However, there
is no wavefront-guided individual treatment to reduce the production of
aberration in SMILE.
Recent clinical studies have
contrasted some pros and cons between SMILE and FS-LASIK to treat myopia[9-13], but there were several different
conclusions regarding the postoperative visual quality between the two
procedures, especially in terms of the biomechanical effects[9,14-16]. Currently, several Meta-analyses
have only investigated the clinical outcome differences in visual acuity and
dry eye between SMILE and FS-LASIK[17-19],
which is insufficient for the evaluation of the two types of surgeries. Therefore,
the purpose of our study was to review mass of relative literatures for
exploring the benefits in visual acuity, aberration, biomechanical effects and
contrast sensitivity between SMILE and FS-LASIK.
MATERIALS
AND METHODS
We conduct the Meta-analysis in
accordance with a prepared protocol, following the generally accepted
recommendations[20-21].
Search Strategy Two reviewers independently searched
PubMed, EMBASE, Cochrane, Web of Science and Chinese databases (WANFANG and
CNKI) up to November 20th, 2016. The search keywords were included: “myopia”, “small-incision
lenticule extraction” or “SMILE” and “FS-LASIK” or “femtosecond” or “laser in
situ keratomileusis”. No date or language restrictions were used for the
research. We scanned the titles and abstracts, retrieved relative full studies
and involved the articles in accordance with our inclusion criteria. Any
disagreement between the reviewers was resolved by discussion.
Inclusion Criteria The following selection criteria was
included: 1) prospective randomized controlled trials (RCTs) and non-randomized
comparative trials; 2) adults with any degree of myopia or myopic astigmatism
without systemic or ocular disease; 3) patients treated with the corneal
surgery (SMILE or FS-LASIK); 4) the follow-up period no less than 3mo; 5)
original clinical articles with independent data was selected.
Outcome Measures The primary outcome parameters were
efficacy, safety, and predictability. The efficacy measure was the proportion
of eyes achieving an uncorrected visual acuity (UCVA) of 20/20 or better. The
safety measures were the percentage of eyes losing one or more lines of best
spectacle corrected distance visual acuity (BSCVA) and the postoperative
spherical equivalent (SE). The refractive SE within ±1.00 diopter (D) of the
target refraction was as the measure of predictability.
The secondary outcomes were
aberration, biomechanical effects and contrast sensitivity. Aberration included
total higher-order aberration (tHOA), spherical aberration, horizontal coma and
vertical coma, and biomechanical effects included corneal hysteresis (CH) and
corneal resistance factor (CRF). The follow-up period ranged from three to six
months, and data were extracted and analyzed from the included studies.
Data Extraction and Quality
Assessment The data extraction and
quality assessment were independently finished by two reviewers, and the
following information was extracted: the first author, design, year, country,
enrolled eyes number, preoperative SE, follow-up time and scores of assessment.
The Jadad scale[22] was used to assess the RCTs, while the
Newcastle-Ottawa scale (NOS)[23] was adopted to evaluate the
cohorts. Randomization, blinding, and participant withdrawal/dropout were the
parameters of the Jadad scale, and the scores of Jadad scale ranged from
minimum of 0 (low quality) and maximum of 5 (high quality). Each one point was
allocated for the parameters of the Jadad scale respectively and additional one
point was obtained when randomization and blinding were appropriate. The NOS
contains the following three main areas of assessment: selection quality,
comparability, and outcome measures. The study was considered high quality when
scoring >3 points in the Jadad scale or scoring >6 points in the NOS.
Statistical Analysis The Meta-analysis was completed with the
RevMan software (version 5.2). The mean difference (MD) was used for continuous
outcomes, and the odds ratio (OR) was calculated for dichotomous outcomes. The
corresponding 95% CI was used for summary estimates, and statistically
significant was a P<0.05.
The Chi-square and I2 statistics
were used to assess heterogeneity. The fixed effect model (FEM) was used
without significant heterogeneity. However, the random effect model (REM) was
used when heterogeneity was obvious (P<0.10 or I2 was >50%).
The robustness of the results was
evaluated with sensitivity analysis, which was performed by excluding the
individual studies one by on to assess its influence on the pooled estimation.
Begg’s and Egger’s tests were adopted to estimate publication bias using STATA[24-25] (version 12.0).
RESULTS
Search Results A total of 201 relative studies were
selected through the electronic databases. After titles and abstracts were
screened, 123 studies were excluded and 19 studies were found ineligible for
inclusion after a systematic review. Finally, 2 RCTs[10,12] and 25 cohorts[7,9,13-16,26-44]
were involved. The reasons to exclude studies were as follows: 2 studies did
not have qualifying interventions, 1 study did not have measurable outcomes, 2
studies were simple letters or commentaries, 3 studies were experiments, 5
studies were duplicates, and 6 studies did not consist with inclusion criteria.
Study Characteristics and
Quality The characteristics and the
quality assessment of the included studies were summarized in Tables 1, 2. A total
of 4223 eyes were included, of which 1928 eyes (45.65%) treated with SMILE and
2295 eyes (54.35%) treated with FS-LASIK. The randomization measures that were
used were inadequate in the RCTs[10,12],
and there was no blinding of the surgeons or patients. When compared with
non-randomized cohort studies, the following factors were not significantly
different between groups within the studies: age, gender, preoperative SE,
aberration, CH or CRF[7,9,13-16,26-44]. Only eleven
studies had six months of follow-up[7,9,14-15,28,30,33,37-39,44].
Therefore, both RCT studies were considered low quality (scoring<3)
according to the standard of Jadad scale, and twenty-four non-randomized
comparative studies scored of high quality (NOS≥6) except for Shen et al[42] 2014 (NOS=5).
Table
1 Characteristics of included studies contrasting SMILE to FS-LASIK mean±SD
|
Study |
Design |
Year |
Country |
SMILE
group |
FS-LASIK
group |
Follow-up (mo) |
Jadad |
NOS |
||
|
Eyes (n) |
Preoperative |
Eyes (n) |
Preoperative |
|||||||
|
Hu et al[26] |
CT
(prospective) |
2013 |
China |
82 |
-4.91±1.29 |
82 |
-6.29±2.37 |
3 |
- |
7 |
|
Hu et al[27] |
CT
(prospective) |
2013 |
China |
83 |
-4.91±1.29 |
94 |
-6.26±2.33 |
3 |
- |
6 |
|
Lin et al[40] |
CT
(prospective) |
2013 |
China |
33 |
-4.81±1.47 |
37 |
-5.56±2.08 |
3 |
- |
7 |
|
Lin et al[13] |
CT
(prospective) |
2014 |
China |
60 |
-5.13±1.75 |
51 |
-5.58±2.41 |
3 |
- |
7 |
|
Denoyer et
al[37] |
CT
(prospective) |
2015 |
France |
30 |
-4.65±2.38 |
30 |
-4.42±1.78 |
6 |
- |
8 |
|
Wang et al[14] |
CT
(retrospective) |
2016 |
China |
50 |
-7.60±1.12 |
56 |
-7.68±1.19 |
3 |
- |
7 |
|
Sefat et
al[41] |
CT
(prospective) |
2016 |
Germany |
43 |
-3.81±0.95 |
26 |
-3.65±1.12 |
3 |
- |
6 |
|
Wu et al[7] |
CT
(prospective) |
2014 |
China |
40 |
-5.71±1.19 |
40 |
-5.80±1.14 |
6 |
- |
8 |
|
Li et al[30] |
CT
(retrospective) |
2014 |
China |
22 |
-4.91±0.90 |
43 |
-5.48±2.09 |
6 |
- |
6 |
|
Li et al[28] |
CT
(prospective) |
2014 |
China |
72 |
-6.04±1.80 |
70 |
-5.94±1.73 |
6 |
- |
8 |
|
Li et al[38] |
CT
(retrospective) |
2016 |
China |
97 |
-5.33±1.46 |
96 |
-5.61±1.75 |
6 |
- |
8 |
|
Xu and
Yang[44] |
CT
(prospective) |
2014 |
China |
81 |
-5.70±1.70 |
97 |
-5.80±2.01 |
6 |
- |
8 |
|
Li et al[29] |
CT
(retrospective) |
2016 |
China |
40 |
-7.89±0.
87 |
40 |
-7.31±0.66 |
3 |
- |
6 |
|
Ye et al[33] |
CT
(retrospective) |
2014 |
China |
170 |
-5.03±1.89 |
88 |
-5.43±2.32 |
6 |
- |
8 |
|
Shen et al[42] |
CT
(retrospective) |
2014 |
China |
17 |
-6.48±1.22 |
17 |
-8.71±2.02 |
3 |
- |
5 |
|
Ang et al[35] |
CT
(prospective) |
2015 |
Singapore |
172 |
-5.71±2.11 |
688 |
-5.73±2.06 |
3 |
- |
7 |
|
Wu and
Wang[16] |
CT (retrospective) |
2016 |
China |
73 |
-5.80±1.35 |
52 |
-5.46±1.08 |
3 |
- |
7 |
|
Li et al[39] |
CT
(retrospective) |
2015 |
China |
55 |
-5.74±1.39 |
51 |
-6.18±1.61 |
6 |
- |
8 |
|
Zhang et
al[34] |
CT
(retrospective) |
2016 |
China |
95 |
-5.34±1.55 |
69 |
-5.01±1.95 |
3 |
- |
6 |
|
Wu and
Wang[43] |
CT
(retrospective) |
2015 |
China |
75 |
-5.49±1.35 |
75 |
-5.56±1.76 |
3 |
- |
8 |
|
Chan et al[36] |
CT
(prospective) |
2016 |
China |
54 |
-5.23±1.96 |
57 |
-5.82±2.60 |
3 |
- |
7 |
|
Qiao et al[31] |
CT
(prospective) |
2015 |
China |
188 |
-5.24±1.85 |
184 |
-5.24±1.72 |
3 |
- |
7 |
|
Wu et al[32] |
CT
(prospective) |
2015 |
China |
34 |
-6.86±0.84 |
29 |
-7.20±0.82 |
3 |
- |
7 |
|
Xia et al[15] |
CT
(prospective) |
2016 |
China |
69 |
-5.04±2.32 |
59 |
-5.13±1.36 |
6 |
- |
8 |
|
Agca et al[9] |
CT
(prospective) |
2014 |
Turkey |
30 |
-3.62±1.79 |
30 |
-3.71±1.83 |
6 |
- |
8 |
|
Liu et al[10] |
RCT |
2016 |
China |
113 |
-5.22±1.70 |
84 |
-5.18±1.93 |
6 |
1 |
- |
|
Ganesh and
Gupta[12] |
RCT |
2014 |
India |
50 |
-4.95±2.09 |
50 |
-3.54±1.26 |
3 |
1 |
- |
SMILE: Small incision lenticule
extraction; FS-LASIK: Femotosecond laser in situ keratomileusis; RCT:
Randomized comparative trial; CT: Comparative trial; SE: Spheriacl equivalent.
Table
2 NOS for non-randomized comparative studies
|
Study |
Selection |
Comparability |
Outcome |
Sum of
score |
|
Hu et al[26] |
3 |
2 |
2 |
7 |
|
Hu et al[27] |
3 |
1 |
2 |
6 |
|
Lin et al[40] |
3 |
2 |
2 |
7 |
|
Lin et al[13] |
3 |
2 |
2 |
7 |
|
Denoyer et
al[37] |
3 |
2 |
3 |
8 |
|
Wang et al[14] |
3 |
2 |
2 |
7 |
|
Sefat et
al[41] |
3 |
1 |
2 |
6 |
|
Wu et al[7] |
3 |
2 |
3 |
8 |
|
Li et al[30] |
3 |
2 |
3 |
8 |
|
Li et al[28] |
3 |
1 |
2 |
6 |
|
Li et al[38] |
3 |
2 |
3 |
8 |
|
Xu and
Yang[44] |
3 |
2 |
3 |
8 |
|
Li et al[29] |
3 |
2 |
3 |
8 |
|
Ye et al[33] |
3 |
1 |
2 |
6 |
|
Shen et al[42] |
3 |
2 |
3 |
8 |
|
Ang et al[35] |
3 |
- |
2 |
5 |
|
Wu and
Wang[16] |
3 |
2 |
2 |
7 |
|
Li et al[39] |
3 |
2 |
2 |
7 |
|
Zhang et
al[34] |
3 |
2 |
3 |
8 |
|
Wu and
Wang[43] |
3 |
1 |
2 |
6 |
|
Chan et al[36] |
3 |
2 |
3 |
8 |
|
Qiao et al[31] |
3 |
2 |
2 |
7 |
|
Wu et al[32] |
3 |
1 |
2 |
6 |
|
Xia et al[15] |
3 |
2 |
3 |
8 |
|
Agca et al[9] |
3 |
2 |
3 |
8 |
The total scores of NOS ranged
from minimum of 0 (low quality) and maximum of 9 (high quality), basing on the
following standards: patient selection methodology (points ranged from 1 to 4),
comparability of the study groups (points ranged from 1 to 2) and outcomes
measures (points ranged from 1 to 3).
Primary Outcomes
Uncorrected visual acuity of
20/20 or better Seven publications
demonstrated percentage of eyes with UCVA of 20/20 or better, and no
significant differences were found between the SMILE and FS-LASIK groups within
6mo (OR 0.77; 95% CI: 0.54, 1.09; P=0.14; Figure 1)[10,12-13,15,31,35-36].
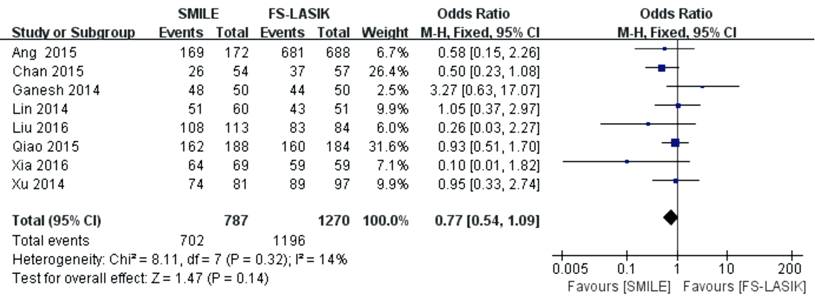
Figure 1 Proportion of eyes
achieving UCVA of 20/20 or better after SMILE versus FS-LASIK within 6mo.
Losing one or more lines of best
spectacle-corrected visual acuity Seven studies reported no significant
differences were found in the percentage of eyes losing one or more lines of
BSCVA between the two groups at the end of follow-up time (OR 1.22; 95% CI:
0.74, 2.03; P=0.43; Figure 2)[10,13,15,27,31,36,44].
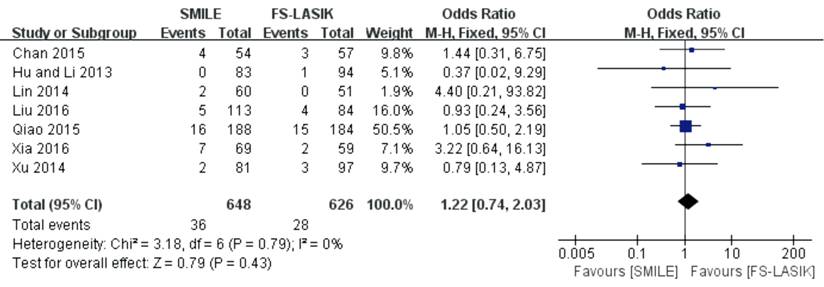
Figure 2 Proportion of eyes
losing one more lines of BSCVA after SMILE versus FS-LASIK within 6mo.
Postoperative mean refractive
spherical equivalent Eleven studies
indicated there was no significant difference of the postoperative mean
refractive SE outcomes between SMILE and FS-LASIK groups (MD 0.00; 95% CI:
-0.04, 0.05; P=0.89; Figure 3)[10,12-13,27,30,34-37,41-42].

Figure 3 Postoperative mean
refractive SE after SMILE versus FS-LASIK within 6mo.
Postoperative refraction within
±1.0 D of the target refraction Six
studies reported no significant difference was found in the postoperative
refraction within ±1.0 D of the target refraction between SMILE and FS-LASIK
groups (OR 0.91; 95% CI: 0.43, 1.93; P=0.80; Figure 4)[10,13,27,31,36,44].
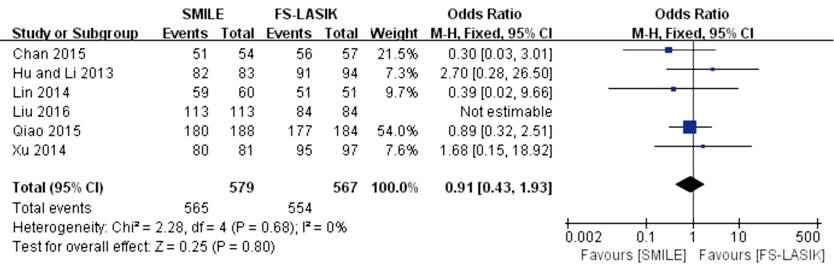
Figure 4 Proportion of eyes with
postoperative refraction within ±1.0 D of target after SMILE versus FS-LASIK
within 6mo.
Secondary
Outcomes
Aberration Ten studies reported the postoperative
aberration at the follow-up times within 6mo. The forest plot showed that tHOA
(MD -0.47; 95% CI: -0.61, -0.33; P<0.00001; Figure 5)[16,26,28,31,33,39,43-44] and spherical
aberration (MD -0.64; 95% CI: -0.88, -0.39; P<0.00001; Figure 6)[10,16,26,28,31,33,39-40,43-44] were lower in the SMILE group than
that in FS-LASIK. No significant difference was found in either the horizontal
coma (MD 0.11; 95% CI: -0.06, 0.28; P=0.19; Figure 7)[10,16,26,28,33,40,43-44] or the vertical
coma (MD -0.10; 95% CI: -0.36, 0.16; P=0.45; Figure 8)[26,28,33,39,43]
between the two groups. A sensitivity analysis was conducted because of the
apparent heterogeneity in the spherical aberration, the horizontal coma and the
vertical coma (the value of I2>50%). In the spherical aberration and
vertical coma outcomes, a apparent heterogeneity (I2>50%) among the
remaining studies didn’t reduce when each study was excluded in turn and the
results of the previous analyses wasn’t changed by any exclusion. Additionally,
the heterogeneity (I2 from 94% to 49%) of postoperative horizontal coma
significantly decreased when the Li et al’s[39] study was
excluded, which did not influence the previous analyses.
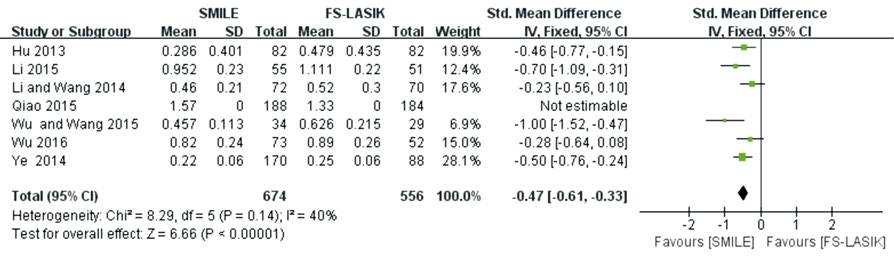
Figure 5 tHOA after SMILE versus
FS-LASIK within 6mo.
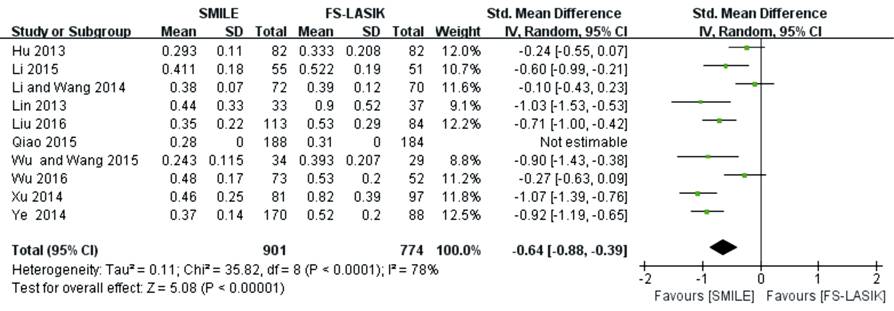
Figure
6 Spherical aberration after SMILE versus FS-LASIK within 6mo.
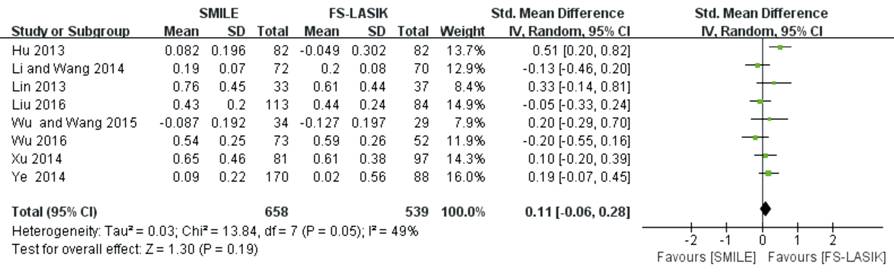
Figure
7 Horizontal coma after SMILE versus FS-LASIK within 6mo.
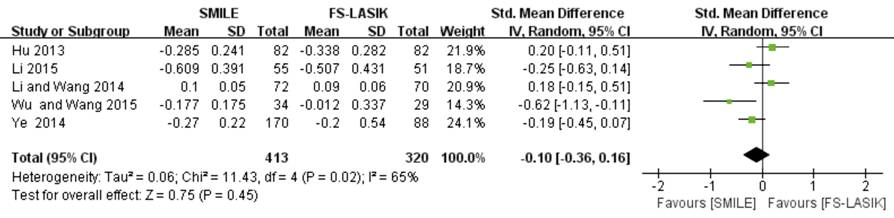
Figure
8 Vertical coma after SMILE versus FS-LASIK within 6mo.
Biomechanical effects Six studies showed significant
differences were found in CH (MD 0.46; 95% CI 0.20, 0.72; P=0.0005; Figure 9)
and CRF (MD 0.67; 95% CI 0.38, 0.96; P<0.00001; Figure 10) between the two
groups. The exclusion of the Agca et al[9] made I2 reduce
from 69% to 40%, but did little influence on the results of CRF (MD 0.48; 95%
CI: 0.21, 1.27; P=0.02).

Figure
9 CH after SMILE versus FS-LASIK within 6mo.
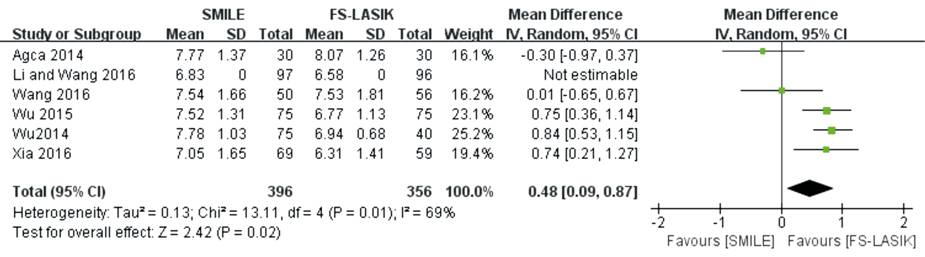
Figure
10 CRF after SMILE versus FS-LASIK within 6mo.
Contrast
sensitivity Four studies reported
changes in contrast sensitivity after SMILE and FS-LASIK[10,12,40,44]. Liu et al[10] indicated that the contrast sensitivity recovered to the
preoperative level later in the SMILE group than that in FS-LASIK. Regarding
the follow-up time, several reports[40,44]
suggested that contrast sensitivity was better in the SMILE group than in
FS-LASIK, particularly at higher spatial frequencies[12].
The changes in contrast sensitivity are presented in Table 3.
Table
3 Changes in contrast sensitivity
|
References |
Method |
Findings |
|
Ganesh et
al[12] |
Measured
using the FACT chart |
At day 1,
contrast sensitivity was better in FS-LASIK group than SMILE group at the
1.5, 3, 6, 12 and 18 cpd , but by 15d and 3mo, contrast sensitivity was
better in SMILE group than FS-LASIK group, particularly at higher spatial
frequencies (18 cpd). |
|
Lin et al[40] |
Measured
using the CGT-1000 |
Contrast
sensitivity was better in SMILE group than FS-LASIK group at the 1.0, 1.7,
and 4.2 cpd without glare and at 2.6, 4.2 and 6.6 cpd with glare after 1mo.
And it was better in SMILE group than FS-LASIK group at the 1.7 and 4.2 cpd
without glare and at 1.7 cpd with glare after 3mo. |
|
Xu and
Yang[44] |
Measured
using the CGT-1000 |
Contrast
sensitivity was better in SMILE group than FS-LASIK group at 6mo
postoperatively. |
|
Liu et al[10] |
Measured
using the CSV-1000E |
In the
SMILE group, the contrast sensitivity at spatial frequencies of 3 cpd and 6
cpd under different lighting conditions recovered to the preoperative level
at 1wk postoperatively, but 12 cpd and 18 cpd recovered to the preoperative
level at 1mo postoperatively. In the FS-LASIK group, the contrast sensitivity
at spatial frequencies of 3, 6, 12, and 18 cpd under different lighting
conditions recovered to the preoperative level at 1wk postoperatively |
Publication Bias No publication bias was apparent using
Begg’s tests (P=0.142 to 0.881) and Egger’s test (P=0.106 to 0.926) (Table 4).
Table
4 The P value of Begg’s tests and Egger’s tests
|
Outcomes |
Begg’s tests |
Egger’s tests |
|
UCVA more
than 20/20 or better |
0.805 |
0.657 |
|
Losing one
or more lines of BSCVA |
0.881 |
0.630 |
|
Mean
refractive SE |
0.784 |
0.926 |
|
Postoperative
refraction within ±1.0 D of target |
0.142 |
0.138 |
|
tHOA |
0.348 |
0.254 |
|
Spherical
aberration |
0.458 |
0.466 |
|
Horizontal
coma |
0.211 |
0.106 |
|
Vertical
coma |
0.327 |
0.428 |
|
CH |
0.624 |
0.900 |
|
CRF |
0.327 |
0.344 |
UCVA: Uncorrected visual acuity;
BSCVA: Best spectacle-corrected visual acuity; SE: Spherical equivalent; tHOA:
Total higher-order aberration; CH: Corneal hysteresis; CRF: Corneal resistance
factor. Publication bias was significant when P≤0.05 using with Begg’s tests
and Egger’s tests.
DISCUSSION
In this Meta-analysis, SMILE
achieved similar efficacy, safety and predictability as FS-LASIK within a 6mo
follow-up time, and the outcomes of horizontal coma and vertical coma were not
significantly different between the two surgeries. Additionally, the increase
in tHOA and spherical aberration in the SMILE group was lower than that in the
FS-LASIK group. In addition, the decrease in the CH and CRF was greater in the
FS-LASIK group compared with the SMILE group, which aided in the investigation
of the fewer impact of biomechanical effects in the SMILE group. Most studies
demonstrated that the contrast sensitivity in SMILE was superior to that in
FS-LASIK. Currently, three Meta-analyses have been published, all of which
focused on efficacy, safety, predictability, dry eye and central corneal
sensitivity. Therefore, our study is the first study to compare the clinical
outcomes of aberration, biomechanical effects and contrast sensitivity between
the SMILE and FS-LASIK techniques using a Meta-analysis with more related
studies included.
Because of the differences in the
details of the reviewed studies, we had difficulty in extracting data and
summarizing the data. We included all data that were consistent with our
inclusion criteria and interpreted the clinical outcomes. When heterogeneity
was observed among the studies, a sensitivity analysis was conducted. In
addition, we used Begg’s rank correction test and Egger’s linear regression
test to determine the publication bias. A major difficulty was the different
measurements of aberration, biomechanical effects and contrast sensitivity.
When detecting aberration and biomechanical effects, these studies used a
different wavefront analyzer or biomechanical instrument, which may account for
the significant difference. Therefore, we selected the parameters of the whole
cornea aberration at 6 mm in diameter and the ocular response analyzer for the
biomechanical effects. Another difficulty was the diverse variation in the
follow-up times. According to our clinical experience and the associated study[18], the parameters of efficacy, safety, predictability,
aberration and biomechanical effects are stable at least three months postoperatively.
Thus, the follow-up time of the Meta-analysis was conducted within 6mo.
This Meta-analysis suggested that
both SMILE and FS-LASIK are effective, safe and predictable. In terms of
efficacy, an examination of the forest plot revealed that no significant
differences were detected between the SMILE and FS-LASIK groups relative to the
proportions with uncorrected distance visual acuities of 20/20 or better. The I2
value of UCVA indicated that heterogeneity was not observed among the studies
and that the results were analyzed using a fixed effects model. Ang et al[35] and Liu et al[10] suggested that the
results of proportions with uncorrected distance visual acuities of 20/20 or
better in the FS-LASIK group were better than in the SMILE group during the 3mo
to 6mo, which may be due to the difference in the healing response between both
procedures. Agca et al[45] reported that eyes treated SMILE
have increased corneal backscatter in the interface 3mo after extracted
lenticule surgery compared with the FS-LASIK procedure.
In terms of safety, the
proportion of eyes losing one or more lines of corrected distance visual acuity
in the SMILE group was similar to those in the FS-LASIK group, which suggested
that both SMILE and FS-LASIK are safe concerning correction of refraction.
In terms of predictability, we
assessed the postoperative mean refractive SE and the proportion of
postoperative refraction within ±1.0 D of the target refraction and no
significant differences were found between the two groups. Additionally, Ganesh
demonstrated that SMILE has greater predictability than FS-LASIK because the
refractive lenticule was cut by a femtosecond laser in SMILE rather than by
lifting the flap and exposing the stroma in FS-LASIK, which may reduce
hydration changes of corneal stroma in SMILE[46-47].
Visual quality is not only visual
acuity but also includes aberration and contrast sensitivity. The refractive
surgery-induced aberration increased after the surgery following up time point
of 6mo. The increasing of tHOA and spherical aberration occurred more in the
FS-LASIK group than in the SMILE group. However, there was not an apparent
difference in horizontal coma and vertical coma between two groups.
Controversially, Wu and Wang[43] reported that the vertical
coma was significantly increased after the SMILE surgery, whereas the
horizontal coma was significantly increased after FS-LASIK surgery. The
postoperative spherical aberration was associated with optical and ablation
zones[48]. There is no transition zone for the SMILE
procedure, and it achieves a larger ablation zone than the FS-LASIK procedures,
which indicated that spherical aberration was lower in SMILE group than that in
FS-LASIK group. With regard to the induction of coma, imbalanced corneal
healing responses and imbalanced optical changes along the axis were involved[49]. Several studies[50-51]
demonstrated that the induction of coma was caused by decentrations after the
SMILE surgery. Whereas, another study[43] reported that
weaker wound healing responses occurred in the SMILE group. In brief, more
research and further studies are needed to investigate the change of aberration
when comparing SMILE with FS-LASIK. Furthermore, the I2 value of spherical
aberration, horizontal coma and vertical coma all showed significant
heterogeneity among studies. Thus, a sensitivity analysis was performed, and
the results were evaluated using a random effects model. For spherical
aberration and vertical coma, the results were analyzed by excluding one study
at a time and demonstrated that the heterogeneity did not change and the
results of the previous analysis were stable. When excluding Li et al[39], the I2 value decreased, but there was no significant
change in the estimated value, which may have been caused by measurement bias.
The refractive surgery-induced
biomechanical effects decreased after surgery demonstrated by six reports[9,14-16,38,43]. The data showed a smaller decrease of CH and CRF in the
SMILE group than in the FS-LASIK group within 6mo after surgery. Nevertheless,
there was significant heterogeneity in CRF by assessing the I2 value.
Sensitivity analysis revealed that the study by Agca et al[9]
was the source of statistical heterogeneity in the Meta-analysis for the CRF,
but the exclusion of the study did not significantly reduce heterogeneity. Agca
et al[9] involved patients with low to moderate myopia,
however, other studies included moderate to high myopia. However, there is no
evidence to identify the correlation between the degree of myopia and the change
of biomechanical effects between two groups. Moreover, Wang et al[14]
reported that there was a significant difference between SMILE and FS-LASIK (P=0.096)
at the 6mo follow-up, although there was no statistically significant
difference in CH. The study of Wang et al[14] can sharply
increase the heterogeneity among these studies (I2 from 0 to 94%) and is the
reason for excluding the study. Several clinical studies[15-16,43] and mathematical analyses[52-53] demonstrated that the SMILE procedure
was superior to the FS-LASIK surgery with respect to the corneal biomechanics.
Otherwise, another study[9] found similar biomechanical
effects between SMILE and FS-LASIK surgery. Biomechanically, the flapless
lenticule extraction technique maximally protects the structural integrity of
the cornea and causes less disruption of the peripheral collagen fibers than
LASIK[7]. Theoretically, the degree of wound repair is
correlated with the inherent strength of the corneal tissues[54].
An in vivo study[55] found that refractive lenticule
extraction might result in less inflammation and early extracellular matrix
deposition than LASIK.
In consideration of contrast
sensitivity, it is known that contrast sensitivity is lower after undergoing
SMILE and FS-LASIK surgery. Several studies[12,40,44] indicated that contrast sensitivity was better in SMILE
group than that in FS-LASIK group except the report of Liu et al[10].
Liu et al[10] reported that the speed of recovery of
contrast sensitivity was due to the different mechanisms of the corneal stromal
wound-healing process after both procedures at high spatial frequencies under
different lighting conditions. Furthermore, several previous studies have
suggested that the decrease in contrast sensitivity was associated with the increase
in HOAs[56-57]. However, Stonecipher and
Kezirian[58] found that there was no relationship between
contrast sensitivity and HOAs.
There are several important
limitations in the Meta-analysis. First, most of the studies were from Asia,
which may cause publishing bias. Second, extracted data of aberration included
various measurements from different wavefront analyzers, which increased the
method bias.
In conclusion, SMILE and FS-LASIK
are comparably safe, effective and predictable when used for the treatment of
myopia. Postoperative aberration and a decrease in biomechanical effects may
occur less frequently after SMILE than after FS-LASIK. Contrast sensitivity was
better in the SMILE group than in the FS-LASIK group 3-6mo postoperatively.
Further randomized, double-blinded, prospective studies with longer follow-up
periods are warranted to provide a better understanding of the benefits of
SMILE and FS-LASIK.
ACKNOWLEDGEMENTS
Conflicts of Interest: Yan H,
None; Gong LY, None; Huang W, None; Peng YL, None.
REFERENCES
1 Vryghem JC, Devogelaere T, Stodulka P. Efficacy, safety, and flap dimensions
of a new femtosecond laser for laser in situ keratomileusis. J Cataract Refract
Surg 2010;36(3):442-448. [CrossRef]
[PubMed]
2 Golas L, Manche EE. Dry eye
after laser in situ keratomileusis with femtosecond laser and mechanical
keratome. J Cataract Refract Surg 2011;37(8):1476-1480. [CrossRef] [PubMed]
3 dos Santos AM, Torricelli AA,
Marino GK, Garcia R, Netto MV, Bechara SJ, Wilson SE. Femtosecond
laser-assisted LASIK flap complications. J Refract Surg 2016;32(1):52-59. [CrossRef] [PubMed]
4 Sekundo W, Kunert KS, Blum M.
Small incision corneal refractive surgery using the small incision lenticule
extraction (SMILE) procedure for the correction of myopia and myopic
astigmatism: results of a 6 month prospective study. Br J Ophthalmol
2011;95(3):335-339. [CrossRef]
[PubMed]
5 Shah R, Shah S, Sengupta S.
Results of small incision lenticule extraction: all-in-one femtosecond laser
refractive surgery. J Cataract Refract Surg 2011;37(1):127-137. [CrossRef] [PubMed]
6 Wei S, Wang Y. Comparison of
corneal sensitivity between FS-LASIK and femtosecond lenticule extraction
(ReLEx flex) or small-incision lenticule extraction (ReLEx smile) for myopic
eyes. Graefes Arch Clin Exp Ophthalmol 2013;251(6):1645-1654. [CrossRef] [PubMed]
7 Wu D, Wang Y, Zhang L, Wei S,
Tang X. Corneal biomechanical effects: small-incision lenticule extraction
versus femtosecond laser-assisted laser in situ keratomileusis. J Cataract
Refract Surg 2014;40(6):954-962. [CrossRef] [PubMed]
8 Demirok A, Ozgurhan EB, Agca A,
Kara N, Bozkurt E, Cankaya KI, Yilmaz OF. Corneal sensation after corneal
refractive surgery with small incision lenticule extraction. Optom Vis Sci
2013;90(10):1040-1047. [CrossRef]
[PubMed]
9 Agca A, Ozgurhan EB, Demirok A,
Bozkurt E, Celik U, Ozkaya A, Cankaya I, Yilmaz OF. Comparison of corneal
hysteresis and corneal resistance factor after small incision lenticule
extraction and femtosecond laser-assisted LASIK: a prospective fellow eye study.
Cont Lens Anterior Eye 2014;37(2):77-80. [CrossRef] [PubMed]
10 Liu M, Chen Y, Wang D, Zhou Y,
Zhang X, He J, Zhang T, Sun Y, Liu Q. Clinical outcomes after SMILE and
femtosecond laser-assisted LASIK for myopia and myopic astigmatism: a
prospective randomized comparative study. Cornea 2016;35(2):210-216. [CrossRef] [PubMed]
11 Pinero DP, Teus MA. Clinical
outcomes of small-incision lenticule extraction and femtosecond laser-assisted
wavefront-guided laser in situ keratomileusis. J Cataract Refract Surg
2016;42(7):1078-1093. [CrossRef]
[PubMed]
12 Ganesh S, Gupta R. Comparison
of visual and refractive outcomes following femtosecond laser-assisted lasik
with smile in patients with myopia or myopic astigmatism. J Refract Surg
2014;30(9):590-596. [CrossRef]
[PubMed]
13 Lin F, Xu Y, Yang Y.
Comparison of the visual results after SMILE and femtosecond laser-assisted
LASIK for myopia. J Refract Surg 2014;30(4):248-254. [CrossRef] [PubMed]
14 Wang B, Zhang Z, Naidu RK, Chu
R, Dai J, Qu X, Yu Z, Zhou H. Comparison of the change in posterior corneal
elevation and corneal biomechanical parameters after small incision lenticule
extraction and femtosecond laser-assisted LASIK for high myopia correction. Cont
Lens Anterior Eye 2016;39(3):191-196. [CrossRef] [PubMed]
15 Xia L, Zhang J, Wu J, Yu K.
Comparison of corneal biological healing after femtosecond LASIK and small
incision lenticule extraction procedure. Curr Eye Res 2016;41(9):1202-1208. [CrossRef] [PubMed]
16 Wu W, Wang Y. Corneal
higher-order aberrations of the anterior surface, posterior surface, and total
cornea after SMILE, FS-LASIK, and FLEx surgeries. Eye Contact Lens
2016;42(6):358-365. [CrossRef]
[PubMed]
17 Kobashi H, Kamiya K, Shimizu
K. Dry eye after small incision lenticule extraction and femtosecond
laser-assisted LASIK: Meta-analysis. Cornea 2017;36(1):85-91. [CrossRef] [PubMed]
18 Shen Z, Shi K, Yu Y, Yu X, Lin
Y, Yao K. Small incision lenticule extraction (SMILE) versus femtosecond
laser-assisted in situ keratomileusis (FS-LASIK) for myopia: a systematic
review and Meta-analysis. PLoS One 2016;11(7):e0158176. [CrossRef] [PMC free article]
[PubMed]
19 Zhang Y, Shen Q, Jia Y, Zhou
D, Zhou J. Clinical outcomes of SMILE and FS-LASIK used to treat myopia: a
Meta-analysis. J Refract Surg 2016;32(4):256-265. [CrossRef] [PubMed]
20 Egger M, Smith GD, Phillips
AN. Meta-analysis: principles and procedures. BMJ 1997;315(7121):1533-1537. [CrossRef]
21 Pogue J, Yusuf S. Overcoming
the limitations of current meta-analysis of randomised controlled trials. Lancet
1998;351(9095):47-52. [CrossRef]
22 Jadad AR, Moore RA, Carroll D,
Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ. Assessing the quality of
reports of randomized clinical trials: is blinding necessary? Control Clin
Trials 1996;17(1):1-12. [CrossRef]
23 Wells GA, Shea B, O'Connell D,
Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for
assessing the quality of non-randomized studies in Meta-analysis. Applied
Engineering in Agriculture 2014;18(6):727-734.
24 Begg CB, Mazumdar M. Operating
characteristics of a rank correlation test for publication bias. Biometrics 1994;50(4):1088-1101. [CrossRef] [PubMed]
25 Egger M, Davey Smith G,
Schneider M, Minder C. Bias in Meta-analysis detected by a simple, graphical
test. BMJ 1997;315(7109):629-634. [CrossRef]
26 Hu YK, Li WJ, Gao XW, Dong J,
Guo YL. Effects of femtosecond laser small incision lenticule extraction on corneal
wavefront aberration in the treatment of myopia. Res Adv Ophtalmol
2013;33(7):651-655.
27 Hu YK, Li WJ, Gao XW, Guo YL,
Dong J. Comparison of small incision lenticule extraction and femtosecond laser
assisted LASIK for myopia. Guoji Yanke Zazhi (Int Eye Sci)
2013;13(10):2074-2077.
28 Li K, Wang YL, Zhang CW, Wu J,
Huang HY. Comparison of SMILE surgery and femotosecond laser LASIK for myopia. Chin
J Ophthalmol Vis Sci 2014;16(8):478-482.
29 Li QH, Li YM, Hou HC, Song XY,
Hu CE, Qi SW. Clinical comparative study of small incision lenticule extraction
and femtosecond laser-assisted LASIK for high myopia. Res Adv Ophthalmol
2016;36(6): 562-565.
30 Li WJ, Hu YK, Gao XW, Cao LP,
Dong J, Guo YL, Cai Y. Evaluation of double-pass optical quality analysis
system between SMILE and femtosecond LASIK for myopia. Guoji Yanke Zazhi (Int
Eye Sci) 2014; 14(11):1971-1974.
31 Qiao BD, Tie B, Zhao H, Ma SG.
Comparison of the efficacy of femtosecond laser small incision extraction and
femtosecond laser assisted LASIK. Chin J Ocul Traum Occupat Eye Dis
2015;37(4):261-265.
32 Wu ZQ, Wang Y, Zhang L, Geng
WL, Jin Y, Zuo T. Wavefront analysis and comparison between small incision
lenticule extraction and femtosecond laser in situ keratomileusis. Zhonghua Yan
Ke Za Zhi 2015;51(3):193-201. [PubMed]
33 Ye M, Liao R, Liu C.
Comparison of higher-order aberrations changes in anterior corneal surface
after four refractive surgeries in myopia. Acta Universitatis Medicinalis Anhui
2014.
34 Zhang JM, Wang Y, Chen XQ, Li
XJ, Xu LL, Dou R. Vector analysis and comprison of small incision lenticule
extraction versus laser in-situ keratomileusis for low to moderate myopiac
astigmatism. Chin J Exp Ophthalmol 2016;34(5):432-437.
35 Ang M, Ho H, Fenwick E,
Lamoureux E, Htoon HM, Koh J, Tan D, Mehta JS. Vision-related quality of life
and visual outcomes after small-incision lenticule extraction and laser in situ
keratomileusis. J Cataract Refract Surg 2015;41(10):2136-2144. [CrossRef] [PubMed]
36 Chan TC, Ng AL, Cheng GP, Wang
Z, Ye C, Woo VC, Tham CC, Jhanji V. Vector analysis of astigmatic correction
after small-incision lenticule extraction and femtosecond-assisted LASIK for
low to moderate myopic astigmatism. Br J Ophthalmol 2016;100(4):553-559. [CrossRef] [PubMed]
37 Denoyer A, Landman E, Trinh L,
Faure JF, Auclin F, Baudouin C. Dry eye disease after refractive surgery
comparative outcomes of small incision lenticule extraction versus LASIK. Ophthalmology
2015;122(4):669-676. [CrossRef]
[PubMed]
38 Li H, Wang Y, Dou R, Wei P,
Zhang J, Zhao W, Li L. Intraocular pressure changes and relationship with
corneal biomechanics after SMILE and FS-LASIK. Invest Ophthalmol Vis Sci 2016;57(10):4180-4186. [CrossRef] [PubMed]
39 Li X, Wang Y, Dou R.
Aberration compensation between anterior and posterior corneal surfaces after
small incision lenticule extraction and femtosecond laser-assisted laser
in-situ keratomileusis. Ophthalmic Physiol Opt 2015;35(5):540-551. [CrossRef] [PubMed]
40 Lin F, Xu Y, Yang Y.
Comparison of the visual results after SMILE and FS-LASIK for treating myopia. J
Refract Surg 2014;30(4):248-254. [CrossRef] [PubMed]
41 Sefat SM, Wiltfang R, Bechmann
M, Mayer WJ, Kampik A, Kook D. Evaluation of changes in human corneas after
femtosecond laser-assisted lasik and small-incision lenticule extraction
(SMILE) using non-contact tonometry and ultra-high-speed camera (Corvis ST). Curr
Eye Res 2016;41(7):917-922. [CrossRef] [PubMed]
42 Shen Y, Chen Z, Knorz MC, Li
M, Zhao J, Zhou X. Comparison of corneal deformation parameters after SMILE,
LASEK, and femtosecond laser-assisted LASIK. J Refract Surg 2014;30(5):310-318.
[CrossRef]
43 Wu W, Wang Y. The correlation
analysis between corneal biomechanical properties and the surgically induced
corneal high-order aberrations after small incision lenticule extraction and
femtosecond laser in situ keratomileusis. J Ophthalmol 2015;2015:758196.
44 Xu Y, Yang YB. A clinical
study of small incision lenticule extraction for the correction of myopia and
astigmatism. University of Zhengjiang 2014.
45 Agca A, Ozgurhan EB, Yildirim
Y, Cankaya KI, Guleryuz NB, Alkin Z, Ozkaya A, Demirok A, Yilmaz OF. Corneal
backscatter analysis by in vivo confocal microscopy: fellow eye comparison of
small incision lenticule extraction and femtosecond laser-assisted LASIK. J
Ophthalmol 2014;2014:265012.
46 Kim WS, Jo JM. Corneal hydration
affects ablation during laser in situ keratomileusis surgery. Cornea 2001;20(4):394-397. [CrossRef] [PubMed]
47 Patel S, Alio JL, Artola A.
Changes in the refractive index of the human corneal stroma during laser in
situ keratomileusis. Effects of exposure time and method used to create the
flap. J Cataract Refract Surg 2008;34(7):1077-1082. [CrossRef] [PubMed]
48 Vega-Estrada A, Alio JL, Arba
Mosquera S, Moreno LJ. Corneal higher order aberrations after LASIK for high
myopia with a fast repetition rate excimer laser, optimized ablation profile,
and femtosecond laser-assisted flap. J Refract Surg 2012;28(10):689-696. [CrossRef] [PubMed]
49 Pallikaris IG, Kymionis GD,
Panagopoulou SI, Siganos CS, Theodorakis MA, Pallikaris AI. Induced optical
aberrations following formation of a laser in situ keratomileusis flap. J
Cataract Refract Surg 2002;28(10):1737-1741. [CrossRef]
50 Kamiya K, Umeda K, Igarashi A,
Ando W, Shimizu K. Factors influencing the changes in coma-like aberrations
after myopic laser in situ keratomileusis. Curr Eye Res 2011;36(10):905-909. [CrossRef] [PubMed]
51 Li M, Zhao J, Miao H, Shen Y,
Sun L, Tian M, Wadium E, Zhou X. Mild decentration measured by a Scheimpflug
camera and its impact on visual quality following SMILE in the early learning
curve. Invest Ophthalmol Vis Sci 2014;55(6):3886-3892. [CrossRef] [PubMed]
52 Reinstein DZ, Archer TJ,
Randleman JB. Mathematical model to compare the relative tensile strength of
the cornea after PRK, LASIK, and small incision lenticule extraction. J Refract
Surg 2013;29(7):454-460. [CrossRef]
[PubMed]
53 Sinha Roy A, Dupps WJ Jr,
Roberts CJ. Comparison of biomechanical effects of small-incision lenticule
extraction and laser in situ keratomileusis: finite-element analysis. J
Cataract Refract Surg 2014;40(6):971-980. [CrossRef] [PubMed]
54 Nishimura M, Yan W, Mukudai Y,
Nakamura S, Nakamasu K, Kawata M, Kawamoto T, Noshiro M, Hamada T, Kato Y. Role
of chondroitin sulfate-hyaluronan interactions in the viscoelastic properties
of extracellular matrices and fluids. Biochim Biophys Acta 1998;1380(1):1-9. [CrossRef]
55 Riau AK, Angunawela RI,
Chaurasia SS, Lee WS, Tan DT, Mehta JS. Early corneal wound healing and
inflammatory responses after refractive lenticule extraction (ReLEx). Invest
Ophthalmol Vis Sci 2011;52(9): 6213-6221. [CrossRef] [PubMed]
56 Holladay JT, Dudeja DR, Chang
J. Functional vision and corneal changes after laser in situ keratomileusis
determined by contrast sensitivity, glare testing, and corneal topography. J
Cataract Refract Surg 1999;25(5):663-669. [CrossRef]
57 Marcos S. Aberrations and
visual performance following standard laser vision correction. J Refract Surg
2001;17(5):S596-601. [PubMed]
58
Stonecipher KG, Kezirian GM. Wavefront-optimized versus wavefront-guided LASIK
for myopic astigmatism with the ALLEGRETTO WAVE: three-month results of a
prospective FDA trial. J Refract Surg 2008; 24(4):S424-430. [PubMed]
--------------------------------------------------------------------------------------------------------------------------------
All rights reserved by Press of International Journal of Ophthalmology (IJO
PRESS)